Not only has the U.K become the first country to vaccinate its citizens, but it has also now become the first country to approve the Oxford/AstraZeneca coronavirus vaccine.
In a statement, the U.K government said the Medicines and Healthcare Products Regulatory Agency (MHRA) had authorized Oxford University/AstraZeneca’s Covid-19 vaccine. Experts at the MHRA are basing the authorization on “rigorous clinical trials and a thorough analysis of the data.”
With this, the United Kingdom has become the first country to authorize the vaccine.
The statement also added that it had met “strict standards of safety, quality, and effectiveness,”
Large Scale Vaccination Programs
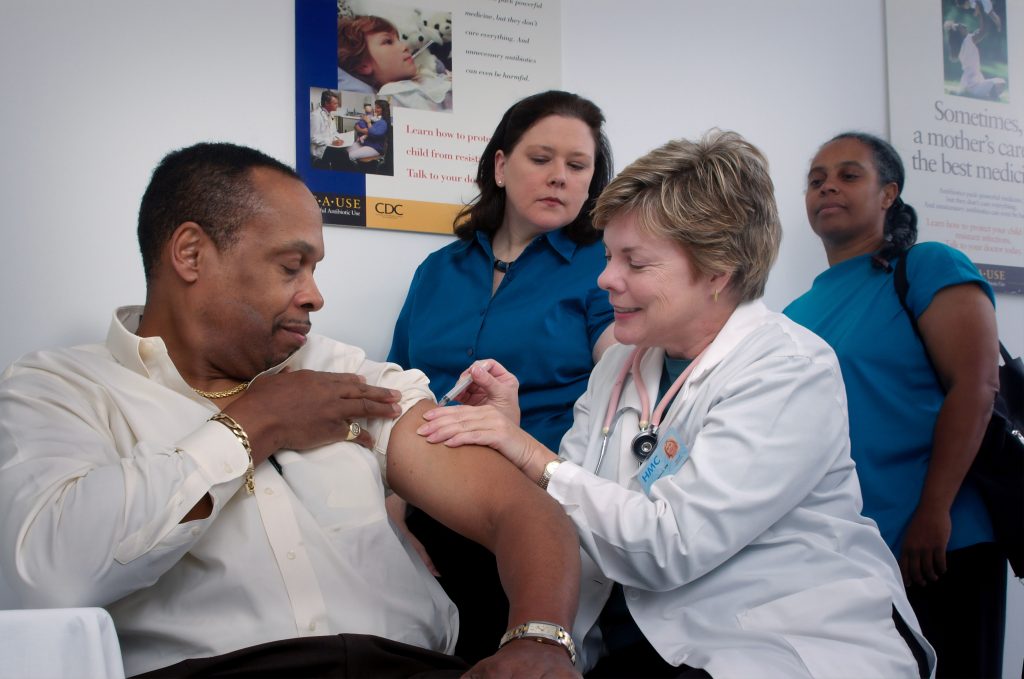 “The NHS has a clear vaccine delivery plan and decades of experience for its delivering large scale vaccination programs. It has already vaccinated hundreds of thousands of patients with the Pfizer/BioNTech vaccine. Its rollout is to continue. Now the NHS will begin putting their extensive preparations into action to roll out the Oxford University/AstraZeneca vaccine.”
“The NHS has a clear vaccine delivery plan and decades of experience for its delivering large scale vaccination programs. It has already vaccinated hundreds of thousands of patients with the Pfizer/BioNTech vaccine. Its rollout is to continue. Now the NHS will begin putting their extensive preparations into action to roll out the Oxford University/AstraZeneca vaccine.”
Health secretary Matt Hancock approved of the critical vaccine on Wednesday. He also says that it means the U.K will be ‘out’ of the coronavirus crisis by the Spring. They also signaled that millions of more people are being put into lockdown.
Given the green light, The Oxford vaccine is now the second one for public roll-out. The USA is the first country to give approval to Pfizer.
It is advised that for long-term protection, one is to get at least two doses. Hancock also revealed that the stocks can be spread more widely than anticipated. The Medicines and Healthcare products Regulatory Agency (MHRA) has also advised. The MHRA is extending the time period between the first jab and the second jab from four weeks to 12 weeks.
Studies have shown that the vaccine has an average efficacy rate of 70 percent. This number rises 90 percent; following half a dose and a full dose.
Also read: Colorado confirms the first case of new COVID-19 strain